Safety evaluation platform for cell therapy products
 Service items
Service items
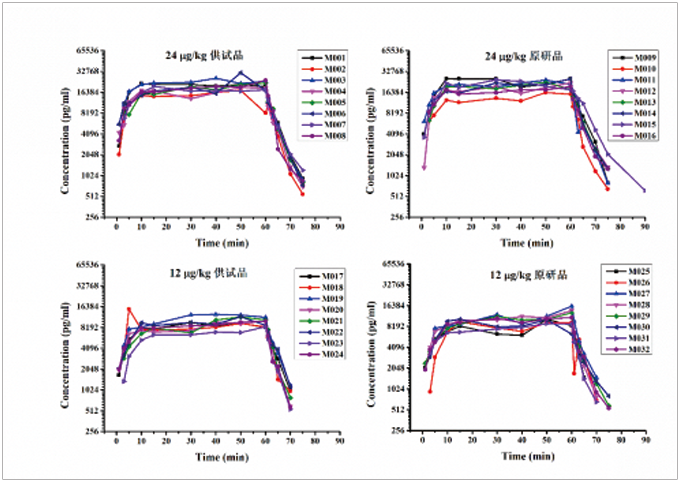
We are capable of conducting safety evaluations for cell therapy products. Currently, we have conducted pharmacodynamic and pharmacokinetic studies on NKT cells, MSCs, and CAR-T cells, as well as a comprehensive set of preclinical safety evaluations.
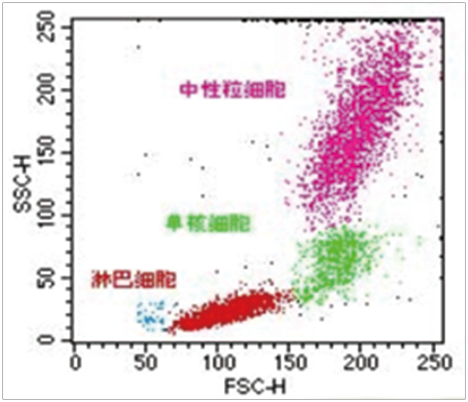
■ Cell count
■ Pharmacodynamic study
■ Single dose toxicity test
■ Repeated dose toxicity test
■ Safety pharmacology test
■ Pharmacokinetic test (including methodological establishment and validation)
■ Local toxicity test
■ Immunogenicity test (including methodological establishment and validation)
■ Tumorigenicity test
■ Tumor forming test
■ Soft agar cloning test
■ Cell count
■ Pharmacodynamic study
■ Single dose toxicity test
■ Repeated dose toxicity test
■ Safety pharmacology test
■ Pharmacokinetic test (including methodological establishment and validation)
■ Local toxicity test
■ Immunogenicity test (including methodological establishment and validation)
■ Tumorigenicity test
■ Tumor forming test
■ Soft agar cloning test
 Company performance - obtaining clinical approval(Only some Company performance are displayed)
Company performance - obtaining clinical approval(Only some Company performance are displayed)
| Drug name | Acceptance No | Indication | Class |
| GKL-006 injection solution | CXSL2300567 | Used to treat irregular primary liver cell carcinoma | 1 |