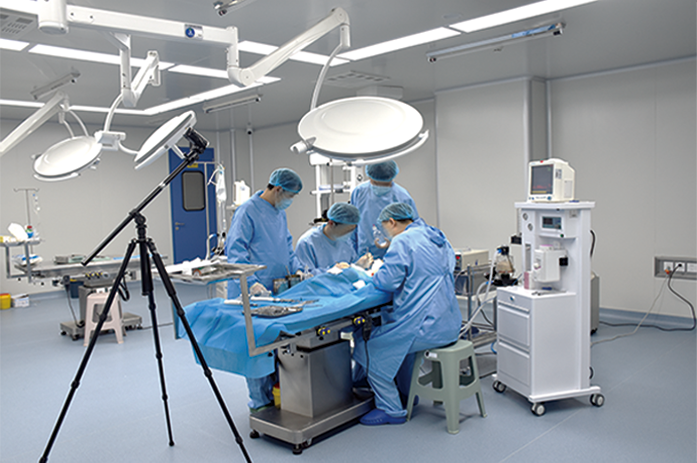
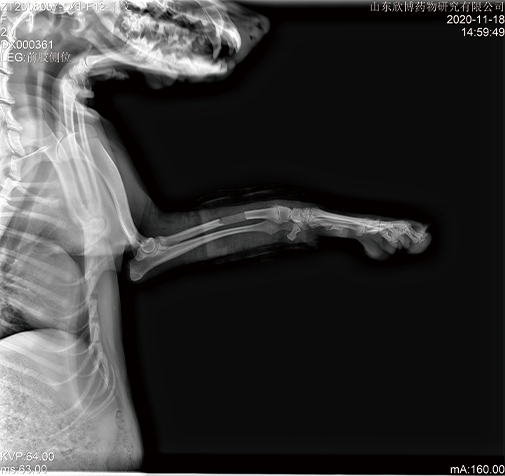
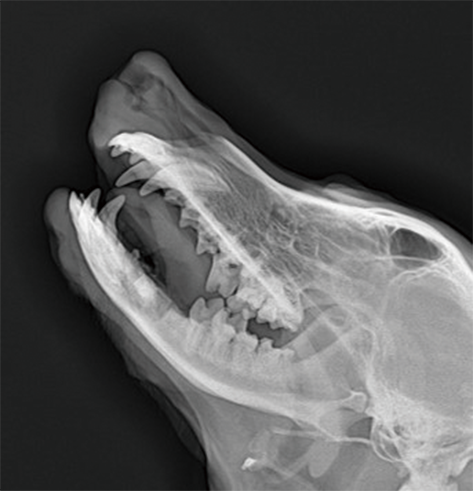
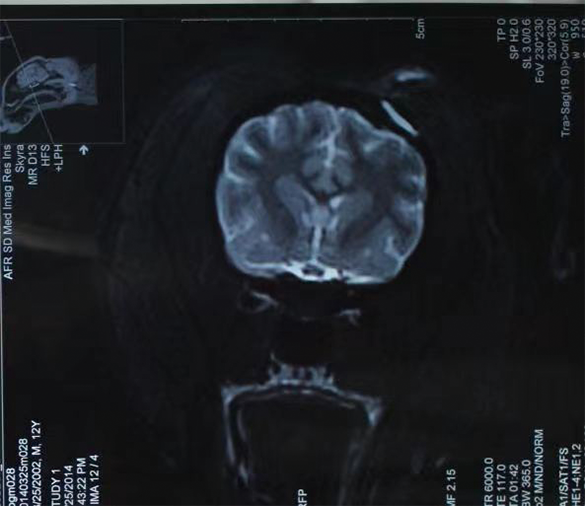
Medical device safety evaluation platform
 Service items
Service items
In the GLP laboratory, non-clinical animal tests for medical devices have been conducted in accordance with regulatory requirements. We have completed multiple studies on the safety and effectiveness of Class III medical devices, including internal hemostatic materials, neurosurgical hemostatic materials, intravascular occlusion or hemostatic materials, dental implant materials, orthopedic implant materials, and novel skin suture materials. These studies have been reviewed and approved.
■ In vitro cytotoxicity test
■ Systemic toxicity test
■ Local reaction test after implantation
■ Stimulation and delayed-type hypersensitivity test
■ Genetic toxicity test
■ Reproductive toxicity test
■ Safety and efficacy trials
■ Non-clinical research on drug-device combination projects
■ In vitro cytotoxicity test
■ Systemic toxicity test
■ Local reaction test after implantation
■ Stimulation and delayed-type hypersensitivity test
■ Genetic toxicity test
■ Reproductive toxicity test
■ Safety and efficacy trials
■ Non-clinical research on drug-device combination projects
 Evaluated varieties
Evaluated varieties
Brain implantation: absorbable hemostatic gauze, absorbable dural sealing medical glue, Fuaile medical glue
>Organ implantation: Kangpaite medical glue, Mefalen sustained-release implant
Dental materials: injectable root filling paste, root canal filling materials
Nasal implantation: biodegradable ear nose styptic sponge
>Muscle pocket implantation: active biological bone
Skin implantation: biological amnion/chorion
>Spine implantation: absorbable hemostatic fluid gelatin
>Subcutaneous implantation: dural medical glue
>Organ implantation: Kangpaite medical glue, Mefalen sustained-release implant
Dental materials: injectable root filling paste, root canal filling materials
Nasal implantation: biodegradable ear nose styptic sponge
>Muscle pocket implantation: active biological bone
Skin implantation: biological amnion/chorion
>Spine implantation: absorbable hemostatic fluid gelatin
>Subcutaneous implantation: dural medical glue
 Company performance - obtaining clinical approval(Only some Company performance are displayed)
Company performance - obtaining clinical approval(Only some Company performance are displayed)
| Drug name | Acceptance No | Indication | Class |
| Use a high -frequency cut operation at one time | National Inspection 20233011227 | This product is used with softening gastrointestinal endoscopy and high -frequency surgical equipment, and uses high -frequency current to cut the tissue in the digestive tract. | Third category |
| Active bone bone | National Inspection 20223131334 | It is used to fill bone defects caused by trauma or surgery and does not affect the stability of the bone structure. | Third category |
| Medical glue | National Inspection 20213020696 | It is used for the closure of surgical incision close to the edge of the skin, including the closure of the minimally invasive intervention surgery puncture. After laparoscopic debridement, spray woun | Third category |
| Absorbing blood vessel seal medical glue | National Inspection 20193020081 | When it is used for blood vessel reconstruction, hemostasis can be assisted through mechanical closure. | Third category |
| Absorbing hardener seal medical glue | National Inspection 20183020031 | It is used for auxiliary seal at the sutures of the hard meningiomy during craniotomy to prevent leakage of the cerebrospinal fluid. During the reconstruction of vascular reconstruction, hemostasis is | Third category |
| Tissue glue | National Inspection 20173024576 | For the treatment of stomach vein rosor embolism. | Third category |
| Tissue adhesion | National Inspection 20173023182 | This product is only used for the use of the body table for local application, which is limited to the combined surgical incision on the edge pair, including the minimally invasive intervention surger | Third category |
| Medical glue | National Inspection 20153021282 | It is used for the closure of surgical incision close to the edge of the skin, including the closure of the minimally invasive intervention surgery puncture. After a laparoscopic debridement, spray wo | Third category |