Drug Regulatory Information | Announcement of the National Medical Products Administration on the Issuance of Work Procedures for the Review and Approval of Conditional Marketing Applications for Drug
2026/04/27
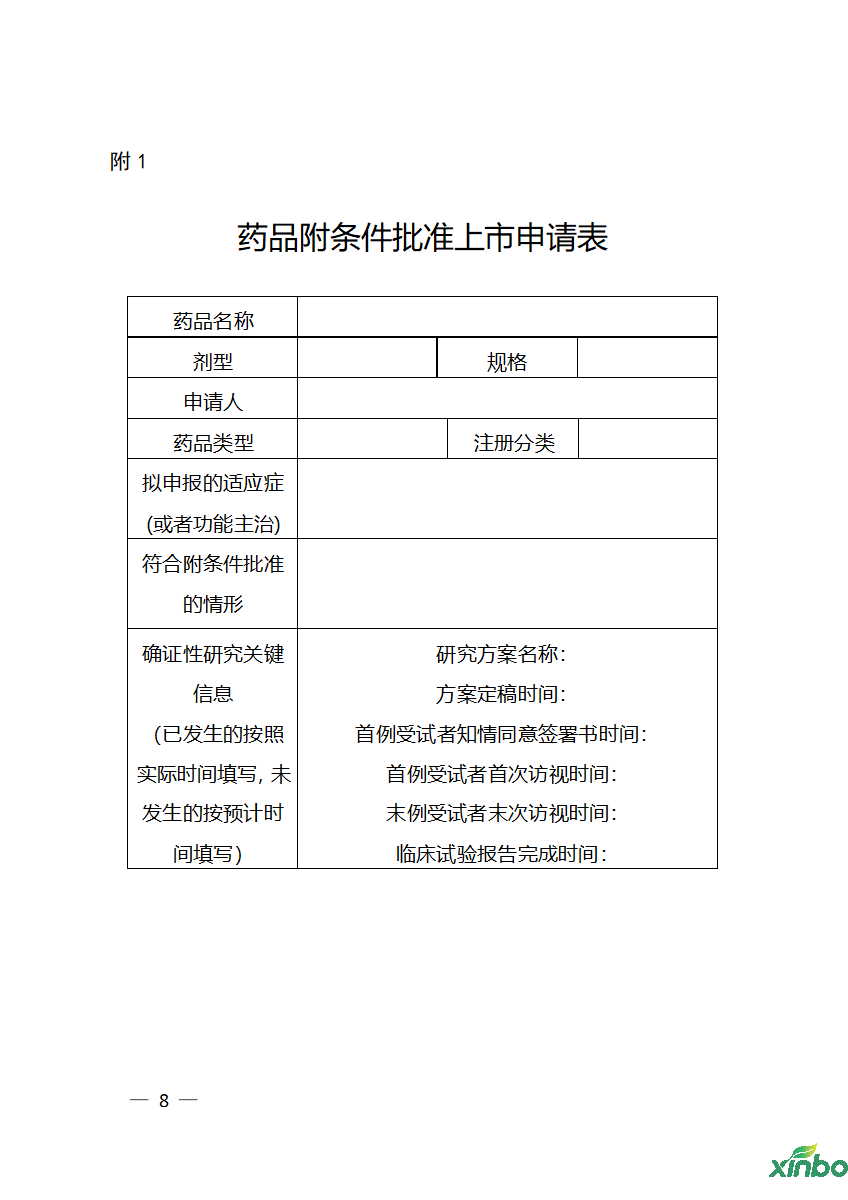
Attachment 1: Work Procedure for Review and Approval of Conditional Approval Application for Drug Marketing










Attachment 2: Policy Interpretation

